
First 7 jurors seated in Trump trial as judge warns former president
Seven Manhattan residents were selected Tuesday afternoon to serve on the jury in former President Donald Trump's criminal trial.
Watch CBS News

Seven Manhattan residents were selected Tuesday afternoon to serve on the jury in former President Donald Trump's criminal trial.

Sen. Bob Menendez heads to trial next month to fight charges that he traded his political influence for cash, gold bars and a luxury car.

The Senate is tasked with the trial after the House impeached Mayorkas earlier this year. Senate Democrats are expected to move to quickly quash the effort.

Trump's allies are encouraging foreign countries to send emissaries to Mar-a-Lago to reconnect ahead of another potential Trump stint in the White House, sources confirmed.

Authorities in Florida said they believe there's a connection between the suspects in the death of a Florida woman and the death of a tow truck driver.

"El Chapo" asked for intervention from the judge in the letter for the "unprecedented discrimination against me."

Both the alleged shooter and the Uber driver appeared to have been targets of scam phone calls, the Clark County Sheriff's Office said.

House Speaker Mike Johnson said Tuesday he won't resign, as another lawmaker calls for him to step down or face an effort to remove him from the top post.
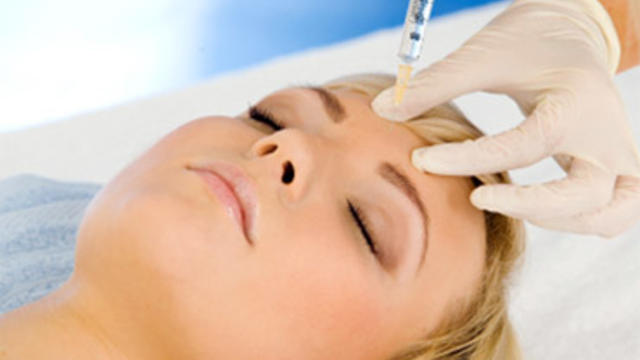
19 people have been stricken — including nine who have been hospitalized — after getting fake or mishandled injections in homes and spas, feds warn.

Strong storms across the central U.S. have caused damage and spawned tornadoes, with more severe storms in the forecast through the night.

Veronica Butler and Jillian Kelley's disappearance last month prompted a murder investigation that has led to four arrests.

A federal appeals court overturned West Virginia's law barring transgender girls from girls' sports teams, finding that it violates Title IX.

The Supreme Court seemed divided over a case challenging the scope of a federal obstruction law that prosecutors have used to charge more than 300 Jan. 6 defendants.

In Game 3 of the 1953 World Series, Erskine struck out 14 hitters for a record that stood until Dodgers ace Sandy Koufax got 15 a decade later.

The $872 million most likely excludes any amount UnitedHealth may have paid to hackers in ransom.
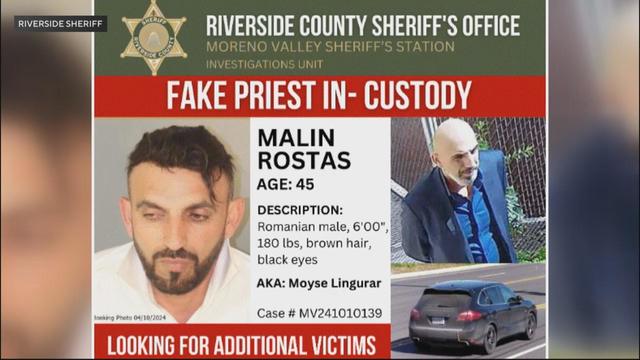
Deputies in Riverside County, California, arrested a man they say posed as a priest and burglarized multiple churches throughout the U.S.

Buzunesh Deba, who lives in the Bronx, became the default winner of the 2014 Boston Marathon in 2016 when the first-place winner was disqualified for doping, but she says she has not yet received her prize money.

"Grief has come to us in waves. Moments of feeling absolutely gutted... then moments of feeling blessed by just getting a moment with her," the couple said.

Seven Manhattan residents were selected Tuesday afternoon to serve on the jury in former President Donald Trump's criminal trial.

Trump's trial will feature a unique cast of characters.

The case stems from a "hush money" payment of $130,000 to adult film star Stormy Daniels in 2016.

President Biden believes painting former President Trump as a "threat" to democracy is a crucial contrast to highlight in his campaign.

The casll came in a joint statement by twelve major news organizations including CBS News.

The Democratic National Committee paid it to firms representing Biden during special counsel Robert Hur's investigation.

Details emerge of Iran's unprecedented direct attack on Israel, and how it was largely thwarted by the U.S. ally's defenses.

President Biden and Israeli Prime Minister Benjamin Netanyahu had a "good conversation," an official said.

President's handling hits new lows; Democrats have grown less supportive of aid to Israel.

The price of gold has been up lately, but by how much. Here's how much gold's price has risen since March 1.

There are multiple types of long-term care insurance to choose from. Learn more about cash benefit policies here.

It can be advantageous for homebuyers to pursue these strategies now before the Fed meets again on April 30.

Republican governors criticized the UAW's efforts as Volkswagen workers in Tennessee are set to start voting on joining the labor union.

The former president's media company announced plans to air news, religious channels and other content.

The $872 million most likely excludes any amount UnitedHealth may have paid to hackers in ransom.

"It must be done," Tesla CEO Elon Musk said in a memo about the layoffs sent to employees on Sunday.

Nike's unitard for female track and field athletes representing the U.S. at the 2024 Paris Olympics is too revealing, critics say.

Receive a $40 Digital Costco Shop Card when you join as a new member at Costco.com when entering PARA24 at checkout.

Do meal kits count as eligible food purchases when it comes to SNAP benefits?

Discover the best riding lawn mowers that'll keep your lawn looking fantastic all year.

Prosecutors and defense attorneys in former President Donald Trump's New York "hush money" criminal trial are getting their first chance to question potential jurors who made it past a first round of screening. CBS News campaign reporter Katrina Kaufman has the latest.

Israeli Prime Minister Benjamin Netanyahu is weighing how to respond to Iran's attack. President Biden told Netanyahu that the U.S. will not participate in a reprisal strike, according to a senior administration official. CBS News foreign correspondent Imtiaz Tyab reports.

House Republicans delivered articles of impeachment against Homeland Security Secretary Alejandro Mayorkas to the Senate on Tuesday. Many Republicans in Congress want to punish Mayorkas for the Biden administration's handling of the U.S.-Mexico border, claiming that the secretary failed to enforce the nation's laws. CBS News' Camilo Montoya-Galvez and Nikole Killion report.

The U.S. Supreme Court on Tuesday appeared divided over a federal obstruction statute used to prosecute hundreds of people who breached the U.S. Capitol building on Jan. 6, 2021. CBS News' Scott MacFarlane breaks down the impact of the case.

In this episode of "Person to Person with Norah O’Donnell," O’Donnell speaks with CVS Health CEO and author Karen Lynch about her life and career.

In this episode of Person to Person with Norah O’Donnell, O’Donnell speaks with author and professor Adam Grant about his newest book, as he discusses unlocking your hidden potential.

In this episode of Person to Person with Norah O’Donnell, O’Donnell speaks with author and professor Arthur Brooks about his partnership with Oprah Winfrey and the key to living a happier life.

In this episode of Person to Person with Norah O’Donnell, O’Donnell speaks with Senator Mitt Romney about his place in the Republican party, his family’s influence and what’s next for him in politics.

In this episode of Person to Person with Norah O’Donnell, O’Donnell speaks with Dolly Parton about her new book on her costumes and clothing and her new rock album.

A federal appeals court overturned West Virginia's law barring transgender girls from girls' sports teams, finding that it violates Title IX.

Recognized as one of CMT's "Next Women of Country," Tanner Adell's career soared after her appearances on Beyoncé's latest album. With a 1,500% spike in Spotify listeners and a new single, "Whiskey Blues," Adell is quickly becoming a standout in the country-pop scene.

Award-winning author Salman Rushdie describes his new memoir as a "reckoning." In 2022, he was stabbed in the neck and abdomen more than a dozen times in western New York and lost sight in his right eye. In Rushdie's new memoir, "Knife: Meditations After an Attempted Murder," he writes about the attack. He and his wife, poet and author Rachel Eliza Griffiths, join "CBS Mornings" for their first joint live interview.

Chicago public school teacher Earnest Horton is the founder of Black Baseball Media, which gives players from predominantly underserved communities access to top-notch facilities and exposure to college scouts. CBS Chicago's Charlie De Mar shares his story on Jackie Robinson Day.
Growing up, Morgan Price said she often felt isolated as one of the few Black gymnasts on her team, a challenge she overcame with the support of her family and, now, her teammates.

Supreme Court justices began hearing arguments Tuesday surrounding a key part of the Jan. 6 prosecutions. The case revolves around the use of a federal obstruction statute. CBS News legal contributor Jessica Levinson joins to break it all down.

Seven jurors have officially been sworn in on Day 2 of former President Donald Trump's New York criminal trial. CBS News chief election and campaign correspondent Robert Costa reports from Manhattan.

The Biden administration is awarding Samsung $6.4 billion to expand American chipmaking. The company will spread the money across at least five facilities in Texas. Sujai Shivakumar, senior fellow at the Center for Strategic and International Studies, joins CBS News to assess the economic and technological impacts.

Israel's military says it will retaliate after Iran and its proxies launched an attack of missiles and drones over the weekend. CBS News foreign correspondent Debora Patta reports. Then, CBS News senior White House correspondent Weijia Jiang explains the U.S.' role in the conflict.

The first criminal trial of a former U.S. president is underway. Jury selection began Monday in Donald Trump's New York "hush money" case. CBS News legal analyst Rikki Klieman joins to discuss.

Spencer, the official mascot of the Boston Marathon, is honored by his community. David Begnaud introduces us to a woman who calls herself a "bad weather friend" – because she's there when you need her most. Plus, more heartwarming stories.

Russ Cook says the scariest part of his run through Africa was "on the back of a motorbike, thinking I was about to die."

A trendsetting third grader creates a school tradition to don dapper outfits on Wednesdays. A retiree makes it her mission to thank those who may be in thankless jobs. Plus, more heartwarming and inspiring stories.

Lyn Story is a retiree whose mission is to be the "bad weather friend," someone who is there for you in a time of need. David Begnaud shows how her huge heart led to life-changing friendships.

Nets star Mikal Bridges fulfills his dream of teaching by working at a school in Brooklyn for the day. A doctor overcomes the odds to help other survivors of catastrophic injuries. Plus, behind the scenes of Drew Barrymore's talk show, and more heartwarming stories.

CBS Reports goes to Illinois, which has one of the highest rates of institutionalization in the country, to understand the challenges families face keeping their developmentally disabled loved ones at home.

As more states legalize gambling, online sportsbooks have spent billions courting the next generation of bettors. And now, as mobile apps offer 24/7 access to placing wagers, addiction groups say more young people are seeking help than ever before. CBS Reports explores what experts say is a hidden epidemic lurking behind a sports betting bonanza that's leaving a trail of broken lives.

In February 2023, a quiet community in Ohio was blindsided by disaster when a train derailed and authorities decided to unleash a plume of toxic smoke in an attempt to avoid an explosion. Days later, residents and the media thought the story was over, but in fact it was just beginning. What unfolded in East Palestine is a cautionary tale for every town and city in America.

In the aftermath of the Supreme Court striking down affirmative action in college admissions, CBS Reports examines the fog of uncertainty for students and administrators who say the decision threatens to unravel decades of progress.

CBS Reports examines the legacy of the U.S. government's terrorist watchlist, 20 years after its inception. In the years since 9/11, the database has grown exponentially to target an estimated 2 million people, while those who believe they were wrongfully added are struggling to clear their names.

CBS News Confirmed examines three viral claims that have emerged so far during Trump's first criminal trial.

Veronica Butler and Jillian Kelley's disappearance last month prompted a murder investigation that has led to four arrests.

Strong storms across the central U.S. have caused damage and spawned tornadoes, with more severe storms in the forecast through the night.

Both the alleged shooter and the Uber driver appeared to have been targets of scam phone calls, the Clark County Sheriff's Office said.

Sen. Bob Menendez heads to trial next month to fight charges that he traded his political influence for cash, gold bars and a luxury car.

Republican governors criticized the UAW's efforts as Volkswagen workers in Tennessee are set to start voting on joining the labor union.

The $872 million most likely excludes any amount UnitedHealth may have paid to hackers in ransom.
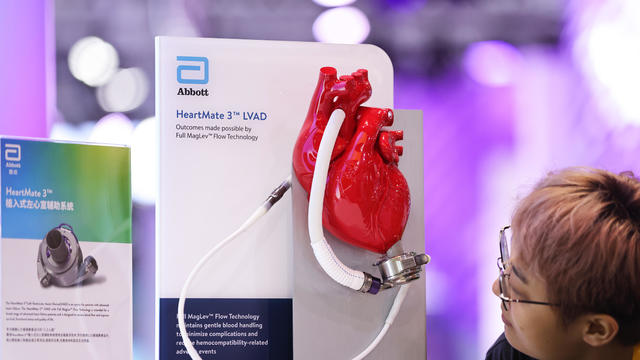
The recall comes years after surgeons say they first noticed problems with the HeartMate II and HeartMate 3, manufactured by Thoratec Corp., a subsidiary of Abbott Laboratories.

The former president's media company announced plans to air news, religious channels and other content.

19 people have been stricken — including nine who have been hospitalized — after getting fake or mishandled injections in homes and spas, feds warn.

CBS News Confirmed examines three viral claims that have emerged so far during Trump's first criminal trial.

Sen. Bob Menendez heads to trial next month to fight charges that he traded his political influence for cash, gold bars and a luxury car.

Trump's allies are encouraging foreign countries to send emissaries to Mar-a-Lago to reconnect ahead of another potential Trump stint in the White House, sources confirmed.

House Speaker Mike Johnson said Tuesday he won't resign, as another lawmaker calls for him to step down or face an effort to remove him from the top post.

The Supreme Court seemed divided over a case challenging the scope of a federal obstruction law that prosecutors have used to charge more than 300 Jan. 6 defendants.

The $872 million most likely excludes any amount UnitedHealth may have paid to hackers in ransom.

The recall comes years after surgeons say they first noticed problems with the HeartMate II and HeartMate 3, manufactured by Thoratec Corp., a subsidiary of Abbott Laboratories.

19 people have been stricken — including nine who have been hospitalized — after getting fake or mishandled injections in homes and spas, feds warn.

"Grief has come to us in waves. Moments of feeling absolutely gutted... then moments of feeling blessed by just getting a moment with her," the couple said.

Bella Hadid raised eyebrows after sharing her elaborate morning routine on TikTok, and other over-the-top celebrity self-care rituals are everywhere. Here's what experts suggest you aim for instead.

Normally parched roads and airport runways in the desert city-state of Dubai were left underwater by an incredibly rare rainstorm.

A whopping 10,000 athletes will carry the torch 3,100 miles over 68 days.

A Sudanese-American family is the first to be reunited in the U.S. after a woman and her sons spent nearly a year stuck in Saudi Arabia.

The 17th-century building's iconic spire, thought to protect the building "against enemy attacks and fires," collapsed among the flames.

The House speaker says he wants to put up separate individual bills on aid for Ukraine, Israel, and Taiwan.

ABBA, Blondie and the Notorious B.I.G. were among the 25 artists selected this year to be added to the Library of Congress' National Recording Registry.

Adell remained tight-lipped about her studio time with Beyoncé, urging fans to use their imagination

ABBA, Blondie and The Notorious B.I.G. are entering America's audio canon.

Recognized as one of CMT's "Next Women of Country," Tanner Adell's career soared after her appearances on Beyoncé's latest album. With a 1,500% spike in Spotify listeners and a new single, "Whiskey Blues," Adell is quickly becoming a standout in the country-pop scene.

A Billy Joel special on CBS and Paramount+ will air again after it was cut off in the middle of the singer's performance of "Piano Man."

The former president's media company announced plans to air news, religious channels and other content.

The Biden administration is awarding Samsung $6.4 billion to expand American chipmaking. The company will spread the money across at least five facilities in Texas. Sujai Shivakumar, senior fellow at the Center for Strategic and International Studies, joins CBS News to assess the economic and technological impacts.

From labor shortages to environmental impacts, farmers are looking to AI to help revolutionize the agriculture industry. One California startup, Farm-ng, is tapping into the power of AI and robotics to perform a wide range of tasks, including seeding, weeding and harvesting.

Roku said Friday a second security breach impacted more than 576,000 accounts after announcing in March that 15,000 accounts had been exposed by a hack. Emma Roth, a writer for The Verge, joins CBS News with more details.

The bill reforms and extends a portion of the Foreign Intelligence Surveillance Act known as Section 702 for a shortened period of two years.

A major global coral bleaching event is occurring for the second time in 10 years, according to the National Oceanic and Atmospheric Administration. Derek Manzello, A coral reef ecologist and NOAA reef watch coordinator, joins CBS News with more.

NASA said it agrees with an independent review board that concluded the project could cost up to $11 billion without major changes.

Only 5 to 6% of plastic waste produced in the U.S. is actually recycled. A new report accuses the plastics industry of a decades-long campaign to "mislead" the public about the viability of recycling.

Mexico City, one of the world's most populated cities with nearly 22 million people, could run out of water in months. Florencia Gonzalez Guerra, an investigative video journalist, joins CBS News to examine the causes behind the crisis.

Greenhouse gas emissions continued increasing in 2023, according to new data from the National Oceanic and Atmospheric Administration. CBS News' Elaine Quijano breaks down the numbers and what they mean for the climate.

Veronica Butler and Jillian Kelley's disappearance last month prompted a murder investigation that has led to four arrests.

Both the alleged shooter and the Uber driver appeared to have been targets of scam phone calls, the Clark County Sheriff's Office said.

Authorities in Florida said they believe there's a connection between the suspects in the death of a Florida woman and the death of a tow truck driver.

Seven Manhattan residents were selected Tuesday afternoon to serve on the jury in former President Donald Trump's criminal trial.

A murder trial starts Tuesday for Karen Read, a Massachusetts woman accused of murdering her boyfriend, who was a police officer, in 2022. Her lawyers say Read is being framed as part of a cover up involving multiple law enforcement agencies, and that someone else is responsible.

NASA confirmed Monday that a mystery object that crashed through the roof of a Naples, Florida home last month was space junk from equipment discarded by the space station.

NASA said it agrees with an independent review board that concluded the project could cost up to $11 billion without major changes.

It was a "bittersweet moment" as United Launch Alliance brought the Delta program to a close.

NASA flight engineers managed to photograph and videotape the moon's shadow on Earth about 260 miles below them.

Millions of Americans poured into the solar eclipse’s path of totality to watch in wonder. The excitement was shared across generations for the rare celestial event that saw watch parties across the country as almost all of the continental U.S. saw at least a partial solar eclipse.

A look back at the esteemed personalities who've left us this year, who'd touched us with their innovation, creativity and humanity.

The Francis Scott Key Bridge in Baltimore collapsed early Tuesday, March 26 after a column was struck by a container ship that reportedly lost power, sending vehicles and people into the Patapsco River.

When Tiffiney Crawford was found dead inside her van, authorities believed she might have taken her own life. But could she shoot herself twice in the head with her non-dominant hand?

We look back at the life and career of the longtime host of "Sunday Morning," and "one of the most enduring and most endearing" people in broadcasting.

Cayley Mandadi's mother and stepfather go to extreme lengths to prove her death was no accident.

Supreme Court justices began hearing arguments Tuesday surrounding a key part of the Jan. 6 prosecutions. The case revolves around the use of a federal obstruction statute. CBS News legal contributor Jessica Levinson joins to break it all down.

House Speaker Mike Johnson is facing pressure from far-right members of his own party, who are threatening to remove him. Meanwhile, articles of impeachment against Homeland Security Secretary Alejandro Mayorkas were delivered to the Senate, where the bid is expected to be quickly dismissed. Scott MacFarlane reports.

Seven jurors have officially been sworn in on Day 2 of former President Donald Trump's New York criminal trial. CBS News chief election and campaign correspondent Robert Costa reports from Manhattan.

ABBA, Blondie and the Notorious B.I.G. were among the 25 artists selected this year to be added to the Library of Congress' National Recording Registry.

Hundreds of unlicensed marijuana farms have cropped up in Maine in recent years, a CBS News investigation has found. Officials believe criminal networks tied to China are behind some of these illegal operations. Nicole Sganga has the story.